- Home >
- Institut Curie News >
- New data on the response of immune sentinels to HIV infection
Different populations of dendritic cells (DC) are present in the tissues in contact with the outside environment where they fulfil the role of sentinels. Their ability to detect pathogenic agents and to trigger specific immune responses means that they are essential for our immune system. Different populations of DC are also present in the blood, where they perform the same function towards the pathogenic agents that reach the bloodstream. Recent studies using large single-cell techniques have helped identify a new and fourth DC sub-population in the human blood, called Axl+ DC.
On HIV infection, the DC in the mucous membranes or the blood, depending on the contamination pathway, would become Trojan horses spreading the virus in the host’s tissue. A few years ago, the team of Philippe Benaroch, Myeloid cells and immunity (Inserm U932 / Université Paris-Cité), showed that among blood DC, the Axl+ DC have the unique ability to bond, replicate and transmit HIV-1*. The way in which different populations of blood DC detect and react to HIV-1 and contribute to antiviral immunity was poorly understood.
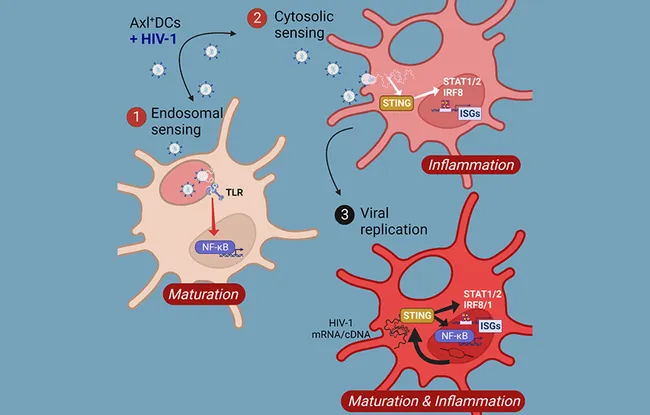
Along with his team, they conducted an in-depth study into the responses elicited by different sub-populations of human DC in the blood. Using transcriptomic analyses of DC sub-populations highly purified from the blood, they showed that the response of Axl+ DC to HIV-1 was a lot stronger and more diverse than that of the other blood DC sub-sets. Furthermore, HIV-1 produces two main transcriptional programs in different Axl+ DC; a program that leads to maturation of DC and activation of T-cells, and a program that leads to antiviral responses (expression of type I human interferons and antiviral genes). Using RNA sequencing in the single-cell on Axl+ DC, the authors showed that the HIV-1 entry pathway could determine the type of responses triggered in these cells and have an impact on the replication of the virus.
Our study was technically challenging, since the cells we studied are very rare, and it is very difficult these days to find infected patients who are not treated. Our collaboration with competent bio-informatics specialists was crucial for implementing the analysis methods that helped us understand how different sub-populations of dendritic cells detect HIV and respond to it. The mechanisms and pathways identified were confirmed by in vitro experiments
Explains Philippe Benaroch.
He also adds that this study provides unique and reliable results on the complex role of dendritic cells during infection by HIV-1. It also indicates that Axl+ DC that have been in contact with HIV could migrate to secondary lymphoid organs. It would be interesting to study the infected DC in tissues such as the lymph nodes, where they would migrate and interact with other immune cells.
Using new technologies such as spatial transcriptomics, it would be possible to actually visualize how these cells direct the immune responses in situ
Philippe Benaroch concludes.