- Home >
- Institut Curie News >
- Response to chemotherapy: finally a biomarker for triple negative breast cancer
In France, triple negative breast cancer (so named because it expresses none of the three markers sought in immunohistology) accounts for around 15% of breast cancer cases, affecting close to 9,000 people each year. Chemotherapy with platinum salts is one of the standard treatments, but not all patients respond positively to it. However, all patients suffer the side effects.
There is currently no biomarker enabling us to target patients who will potentially respond to the treatment. Hope is emerging through the work of Institut Curie researchers, including Dr. Elisabetta Marangoni, deputy manager of the In Vivo Preclinical Experiments platform (CurieCoreTech/Translational research department), Dr. Marc-Henri Stern, head of the DNA Repair and Uveal Melanoma team (Inserm U830/Université Paris Cité) and Dr. Ivan Bièche, practitioner in the Genetics department and the team of Jos Jonkers at the Netherlands Cancer Institute (NKI).
The BRCA1 and BRCA2 mutations are not sufficient
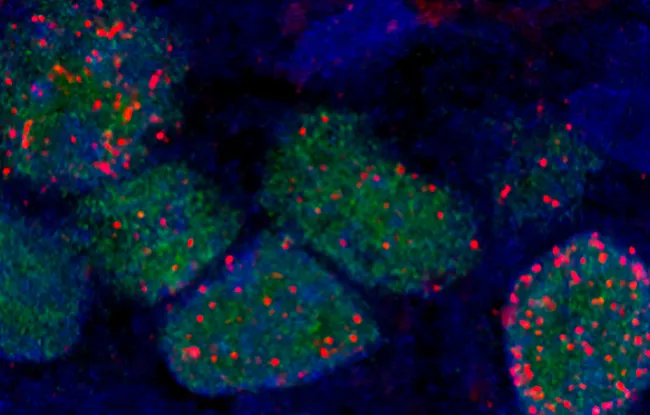
Scientists have started to look at the homologous recombination deficiency (HRD). Cells with this deficiency cannot repair their DNA properly when a break affects both strands. HRD is in fact targeted by platinum salts, as they cause double-strand breaks and the tumor cells die, because they are unable to repair them.
In preclinical models, established based on 55 patient tumors, researchers assessed the correlation between the response to chemotherapy with platinum salts and various potential biomarkers linked to HRD. These include mutations in the BRCA1 or 2 genes or methylation of BRCA1, DNA alterations often causing the HRD. But although they indicate a trend, they are not statistically significant.
This part of the study, however, enabled us to discover that methylation of BRCA1 must be complete for the tumor to respond to platinum salts, which explains the resistance of certain cancers in which BRCA1 is only partially methylated
Explains Dr. Elisabetta Marangoni.
The solution: shallow whole genome sequencing
Another biomarker has nonetheless yielded interesting results, namely the genomic signature of homologous recombination. This involves establishing the genomic profile of the tumor using a technique developed by Dr. Marc-Henri Stern, at Institut Curie, namely shallow whole genome sequencing of the tumor’s DNA.
Using this rapid, inexpensive and easy-to-use technique, we can find out whether a tumor has an HRD or non-HRD profile
Clarifies Dr. Elisabetta Marangoni.
And this time the statistical correlation is clear, since the presence of an HRD profile predicts a response to chemotherapy for 93% of tumors, whereas its absence predicts resistance to chemotherapy in 65% of cases.
Continuing their investigations, the researchers also identified mutations of two other genes (XRCC3 and ORC1), also involved in DNA repair, explaining the response of certain tumors to platinum salts.
To ensure that patients responding to treatment are not excluded, and that those who will not respond are not offered treatment, thus sparing them the side effects of this chemotherapy, one solution would be to take into account the homologous recombination deficiency overall, using shallow whole genome sequencing, without necessarily identifying the mutation causing it
Concludes Dr. Elisabetta Marangoni.
This approach now awaits validation in a clinical setting... Institut Curie’s teams are ready for the challenge.
Reference: P. ter Brugge et al., Homologous Recombination Deficiency derived from Whole-Genome Sequencing predicts Platinum Response in triple-negative Breast Cancers, Nature Communications (2023). DOI: 10.1038/s41467-023-37537-2