- Home >
- Institut Curie News >
- The unsuspected powers of intestinal epithelial cells on the microbiota in the early stages of life
Immune cells play a key role in regulating the interactions between the microbiota and the intestine, and therefore in the proper functioning of the latter. But how does this regulation take place in the early life stages, when the immune system is still immature? A multidisciplinary team from Institut Curie, Inserm, and CNRS unveils previously unknown mechanisms in the journal Science on April 2, 2026.
The links between the intestine and the microbiota—a community of billions of bacteria, viruses, yeasts and other fungi that live in the digestive system – are widely studied and scientists have shown that this relationship is based in particular on the action of the immune system.
However, these links are significantly less well established for the early stages of life, when the immune system is not fully operational. "However, it is precisely during these early stages that the interactions that will shape intestinal function take place," recalls Dr. Pedro Hernandez Cerda, researcher at Inserm and team leader at Institut Curie, within the Genetics and Developmental Biology Unit (Institut Curie, Inserm, CNRS).
To fill this gap, his team joined forces with a dozen other laboratories and studied intestinal development of zebrafish. "One day (only) after hatching, these animals swim with their mouths open," explains Pedro Hernandez Cerda. "They are therefore exposed to the microorganisms of their environment while their immune system is immature, which makes them an ideal model for this subject."
An immune messenger at the heart of the early relationship between the intestine and microbiota
For the first time, scientists have shown that enteroendocrine cells, specialized epithelial cells of the intestine, produce an immunity molecule called interleukin-22 or IL-22, which was previously thought to be the exclusive domain of lymphocytes—cells of the immune system. The team went even further by revealing that the synthesis of this molecule is triggered by the microbiota itself (via the production of the metabolite tryptophan) and that IL-22 in turn shapes the microbiota, by promoting the expression of antimicrobial genes in intestinal epithelial cells.
"It is therefore a self-reinforcing cycle: the microbiota appears to exploit its host, via IL-22, to shape its own composition, which in turn influences intestinal function, summarizes Pedro Hernandez Cerda. We have further shown that this cycle promotes intestinal motility and that ghrelin, a hormone known for its role in appetite, helps counteract the effects of an IL-22 deficiency on this motility." In young animals deficient in IL-22, the researchers observed slower intestinal transit as well as reduced ghrelin levels.
Enteroendocrine cells, specialists in multitasking?
"This circuit involving enteroendocrine cells seems to act specifically at the beginning of life. Thus, it could constitute a therapeutic target for certain disorders of motility or intestinal inflammation in the early stages of life in mammals and therefore in humans, estimates the researcher. Our findings also highlight the importance of intestinal epithelial cells, such as enteroendocrine cells, whose capacities, especially in terms of immunity, have perhaps been underestimated so far."
Building on these results, the scientists now intend to further study these cells, in particular their potential role in intestinal regeneration in zebrafish, if any. "We have noticed the high plasticity of these cells and their crucial role in intestinal development, and will now evaluate them under even more extreme conditions, for example, following severe intestinal," points out Dr. Pedro Hernandez Cerda.
Who knows if they won't then reveal astonishing new powers?

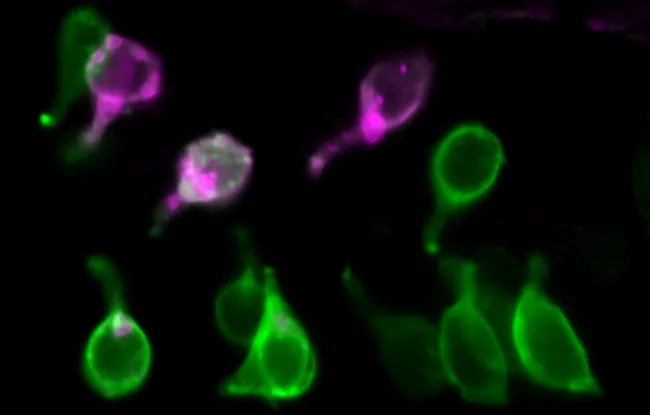
Live zebrafish larva showing intestinal epithelial cells in green and cells expressing IL-22 in red (copyright: Institut Curie / team P. Hernandez Cerda)
Reference : IL-22 from enteroendocrine cells promotes early-life gut motility in zebrafish through the microbiota |
Soraya Rabahi, Lucie Maurin, Emiliano Marachlian, Fabian Guendel, Aya Mikdache, Keinis Quintero-Castillo, Vincenzo Di Donato, Jessica Riou-Ramon, Akshai Janardhana Kurup, Yazan Salloum, Gwendoline Gros, Patricia Diabangouaya, Camila Garcia-Baudino, Ignacio Medina-Yanez, Pascal Hersen, Alvaro Banderas, Jean-Pierre Levraud, Georges Lutfalla, Filippo Del Bene, Carmen G. Feijoo, Pedro P. Hernandez. Science, published on April 2, 2026. N°DOI 10.1126/science.adr1707XXX